ISTO researcher Giada Iacono Marziano and her team publish in Nature Communications a major discovery on the magmatic mechanisms driving the formation of world-class deposits of energy transition metals.
Magmatic sulfide deposits provide a significant proportion of the world’s PGM (over 95%) and nickel (over 40%) production, and are important sources of copper and cobalt. An economically viable deposit of these metals must be discovered each year to meet the growing demand of the electric vehicle battery industry. The scientific community is working to establish conceptual models to better understand the phenomena behind such deposits, and thus be able to predict their distribution and location.
In mafic and ultramafic magmas¹, metal-rich sulphide liquids can form during the transit of the magma to the surface, and accumulate locally forming deposits. These deposits are exploitable provided that they represent a high concentration of critical metals, such as platinoids, nickel, copper, or cobalt. Researchers have conventionally focused on the study of equilibria between the sulphide liquid and the silicate liquid to understand the factors of enrichment in these critical metals in magmatic sulphides. This research has yielded relatively few insights into understanding and predicting why some deposits are vastly superior to others in terms of ore quantity and/or metal enrichment.
Researchers from ISTO, Orleans, in collaboration with a group from the Australian CSIRO, show that magmatic degassing, involving a third phase, the fluid phase, is a phenomenon capable of increasing tenfold (i) the efficiency of the accumulation of sulphide liquids, and (ii) the enrichment in critical metals of these sulphides. Moreover, if the magmatic degassing is pushed, it is even able to form platinoid nuggets directly in the magma.
It is by experimentation in magmatic conditions (high temperature – high pressure) that this three-body mechanics could be highlighted. The synthetic magmas produced in Orleans illustrate the association between gas bubbles (fluid phase) and sulphide liquid (see figure). This association is preserved in the world-class Noril’sk deposit in Siberia, and suggests that the presence of a fluid phase in a magma represents the critical parameter for the formation of the richest magmatic sulfide deposits.
Reference : Iacono-Marziano, G., Le Vaillant, M., Godel, B.M. et al. The critical role of magma degassing in sulphide melt mobility and metal enrichment. Nat Commun 13, 2359 (2022). https://doi.org/10.1038/s41467-022-30107-y
_
1 Magmas that are among the hottest on earth.
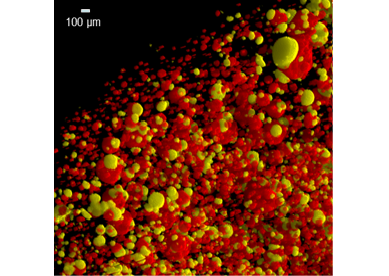
3D distribution of sulfide liquid droplets and fluid phase bubbles in an experimental sample, shown by high-resolution X-ray tomography (HRXCT). The sulfide liquid is shown in yellow and the fluid phase bubbles in red.


